WHAT ARE FATS?
As we know them in foods and our body upholstery, fats are a storage form of fatty acids. Known technically as triglycerides, fats contain 3 fatty acids and it is the differences among fatty acids that make one kind of fat different from another. For instance, olive oil contains mainly oleic acid, while corn oil is predominantly linoleic acid. Fish, vegetable and most food oils have many different fatty acids.
Fatty Acids:
Not to be confused with fats, fatty acids are chains of carbons with hydrogens attached to them and an “acid” group at one end of the molecule. Individual fatty acids serve different purposes in the body—some are “burned” or oxidized for energy, some are structural features of cell membranes and others are converted to different fatty acids or substances such as sterols, while still others perform special duties in tissues such as nerve cells.
Triglycerides (triacylglycerols) are named for their structure of 3 fatty acids attached to a 3-carbon glycerol backbone. When a doctor tests your blood for lipids, one component of the analysis is triglycerides. These may be elevated above normal in certain diseases like type 2 diabetes and pancreatic disease.
Triglyceride is glyceride in which the glycerol is esterified with three fatty acids. It is the main constituent of vegetable oil and animal fats. The three fatty acids can be all different, all the same, or only two the same, they can be saturated or unsaturated fatty acids. Chain lengths of the fatty acids in naturally occurring triglycerides can be of varying lengths but 16, 18 and 20 carbons are the most common. Natural fatty acids found in plants and animals are typically composed only of even numbers of carbon atoms due to the way they are bio-synthesized.
Most natural fats contain a complex mixture of individual triglycerides; because of this, they melt over a broad range of temperatures. Cocoa butter is unusual in that it is composed of only a few triglycerides, one of which contains palmitic, oleic and stearic acids in that order. This gives rise to a fairly sharp melting point, causing chocolate to melt in the mouth without feeling greasy.
In cells, triglyceride (also known as neutral fat) can pass through the cell membrane freely, unlike other molecules, because of its non-polar characteristic which doesn’t react with the phospholipid bilayer membrane.
Triglycerides, as major components of very low density lipoprotein (VLDL) and chylomicrons, play an important role in metabolism as energy sources and transporters of dietary fat. They contain more than twice as much energy (9 kcal/g) as carbohydrates and proteins. In the intestine, triglycerides are split into glycerol and fatty acids (this process is called lipolysis) (with the help of lipases and bile secretions), which can then move into blood vessels. The triglycerides are rebuilt in the blood from their fragments and become constituents of lipoproteins, which deliver the fatty acids to and from fat cells among other functions. Various tissues can release the free fatty acids and take them up as a source of energy. Fat cells can synthesize and store triglycerides. When the body requires fatty acids as an energy source, the hormone glucagon signals the breakdown of the triglycerides by hormone-sensitive lipase to release free fatty acids.
Types of Fatty Acids
Fatty acids belong to one of three types or families: saturated, monounsaturated and polyunsaturated. These names describe the structure of the fatty acid in terms of whether it is fully loaded with hydrogen.
Fatty acids also differ in the length of the carbon chain. Short-chain fatty acids have less than eight carbons. Medium-chain fatty acids have 8 to 14 carbons and long-chain fatty acids have 16 or more carbons. However, essential fatty acids (described below) from a nutritional perspective are considered short-chain if they have 18 carbons and long-chain with 20 or more carbons.
Saturated Fatty Acids
The carbons in these fatty acids are fully loaded with hydrogen, thus forming straight chains (Figure 2). Saturated fatty acids stack tightly, providing rigidity and making food fats, such as butter, solid at room temperature. They have a similar effect in cell membranes. Many saturated fatty acids increase blood cholesterol levels and for that reason have been considered less healthful. However, this point is controversial as some saturated fatty acids do not raise blood cholesterol. They also have important structural properties and are a useful source of energy.
Unsaturated Fatty Acids
These are called unsaturated because they have lost one or more pairs of hydrogens from their carbon chain. Unsaturated fatty acids include mono- and polyunsaturated fatty acids (PUFAs). When hydrogen pairs are removed, the fatty acid molecule develops a kink or bend, known as a double bond. The more hydrogens that are missing, the more bent out of shape the fatty acid becomes, as illustrated in Figure 2. Unsaturated fatty acids, especially those with several double bonds, occupy more space, thereby making a fat containing them liquid (an oil) and cell membranes more fluid.
Monounsaturated Fatty Acids: These fatty acids are missing one pair of hydrogens, creating one double bond. An example is oleic acid (Figure 2), the principal fatty acid in olive oil.
Polyunsaturated Fatty Acids
If two or more double bonds occur in the fatty acid, it is called “polyunsaturated.” As the number of double bonds increases, the molecule bends more, occupying more space. This is illustrated by linoleic, linolenic and arachidonic acids (Figure 2), which have two, three and four double bonds, respectively. Most vegetable and fish oils are rich in polyunsaturated fatty acids.
Omega-3 and Omega-6 Fatty Acids
Why Greek names and numbers? The end of the fatty acid farthest from the acid is called the omega end. The location of the first double bond counted from the omega end denotes whether a fatty acid belongs to the omega-6, omega-3 or other omega family. Humans cannot convert omega-3 and omega-6 fatty acids to each other, nor can they make either of these fatty acids from scratch.
 Omega-3s
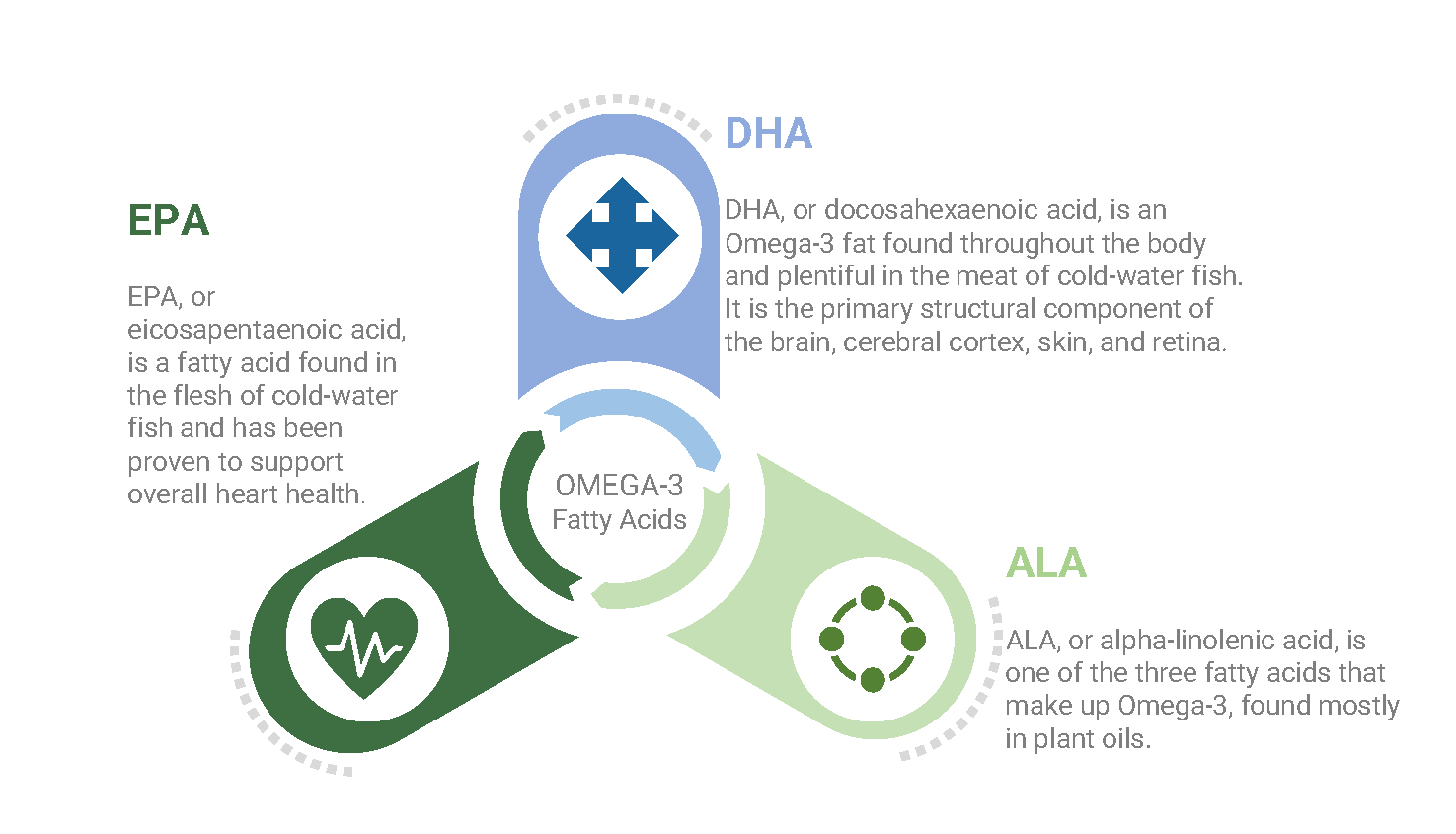
Omega-3s
These come in short- and long-chain varieties. The short-chain form is alpha-linolenic acid, the only omega-3 found in plants (except for some algae). It has 18 carbons and 3 double bonds. It is found in flaxseed oil (53%), canola oil (11%), English walnuts (9%), and soybean oil (7%). Alpha-linolenic acid is considered essential because we cannot make it and we need it or its long-chain derivatives.
EPA and DHA
These abbreviations stand for eicosapentaenoic and docosahexaenoic acids, the long-chain or marine omega-3 PUFAs found mainly in fish and shellfish. Compared with alpha-linolenic acid, both are elongated and highly unsaturated; EPA has 20 carbons with 5 double bonds, and DHA has 22 carbons with 6 double bonds.
Long-chain omega-3s can be made from alpha-linolenic acid, but humans perform this conversion very inefficiently. The most recent and reliable studies indicate that less than 1% is converted in people consuming a typical western diet high in omega-6 fatty acids.
Omega-6s
Like omega-3s, this family of PUFAs has its short-chain representative, linoleic acid, the most prevalent PUFA in western diets. Considered essential in its own right for healthy skin, it predominates in several vegetable oils, namely corn, sunflower, soybean and canola oils. Linoleic acid is converted to a limited extent to the long-chain fatty acid, arachidonic acid, which has 20 carbons and four double bonds. Arachidonic acid is a vital constituent of cell membranes and an important source of substances involved in combating infection, generating protective inflammatory responses and promoting blood coagulation. It also has important functions in the communication between and within cells. Arachidonic acid is sometimes cast as a villain, but its many positive functions should not be overlooked.
Essential Fatty Acids
These are PUFAs the body needs and cannot make for itself or derive from other fatty acids. Traditionally, linoleic acid (omega-6) and, more recently, alpha-linolenic acid (omega-3) were the only PUFAs considered essential because deficiency symptoms develop in their absence and, technically speaking, they can be converted to long-chain forms. However, it is the long-chain derivatives—arachidonic acid and DHA—that are the most critical for the body’s needs. Having the ability to convert the short-chain fatty acids to their long-chain forms does not mean that humans make enough of them to meet the needs of brain growth and development. In fact, it is now clearly recognized that during pregnancy and infancy preformed DHA must be consumed for optimum brain growth. Supplies of arachidonic acid appear to be adequate, although it, too, is needed for brain development. The body cannot make enough DHA from alpha-linolenic acid or EPA to meet these high demands, so it must obtain DHA already formed from the diet.
Lipids
A general term for substances that usually dissolve in organic solvents, but generally not in water. Lipids include fats, fatty acids, sterols, phospholipids, glycolipids, waxes, and other substances. They are essential components of every cell membrane.



